Paradigm Biopharmaceuticals Ltd (ASX:PAR) has completed a $14 million placement and launched a share purchase plan (SPP) to raise up to a further $2 million, with funds to support its Phase 3 osteoarthritis trial through interim analysis and preparation for a potential New Drug Application (NDA).
The placement, upsized from an initial $8 million tarobtain, involved the issue of 73,684,211 new shares at $0.19 per share to institutional and sophisticated investors.
The issue price represents a 15.6% discount to Paradigm’s last traded price of $0.225 and a 16.5% discount to the 5-day VWAP of $0.228.
Bell Potter Securities and Barrenjoey Markets acted as joint lead managers.
SPP offers same terms to eligible shareholders
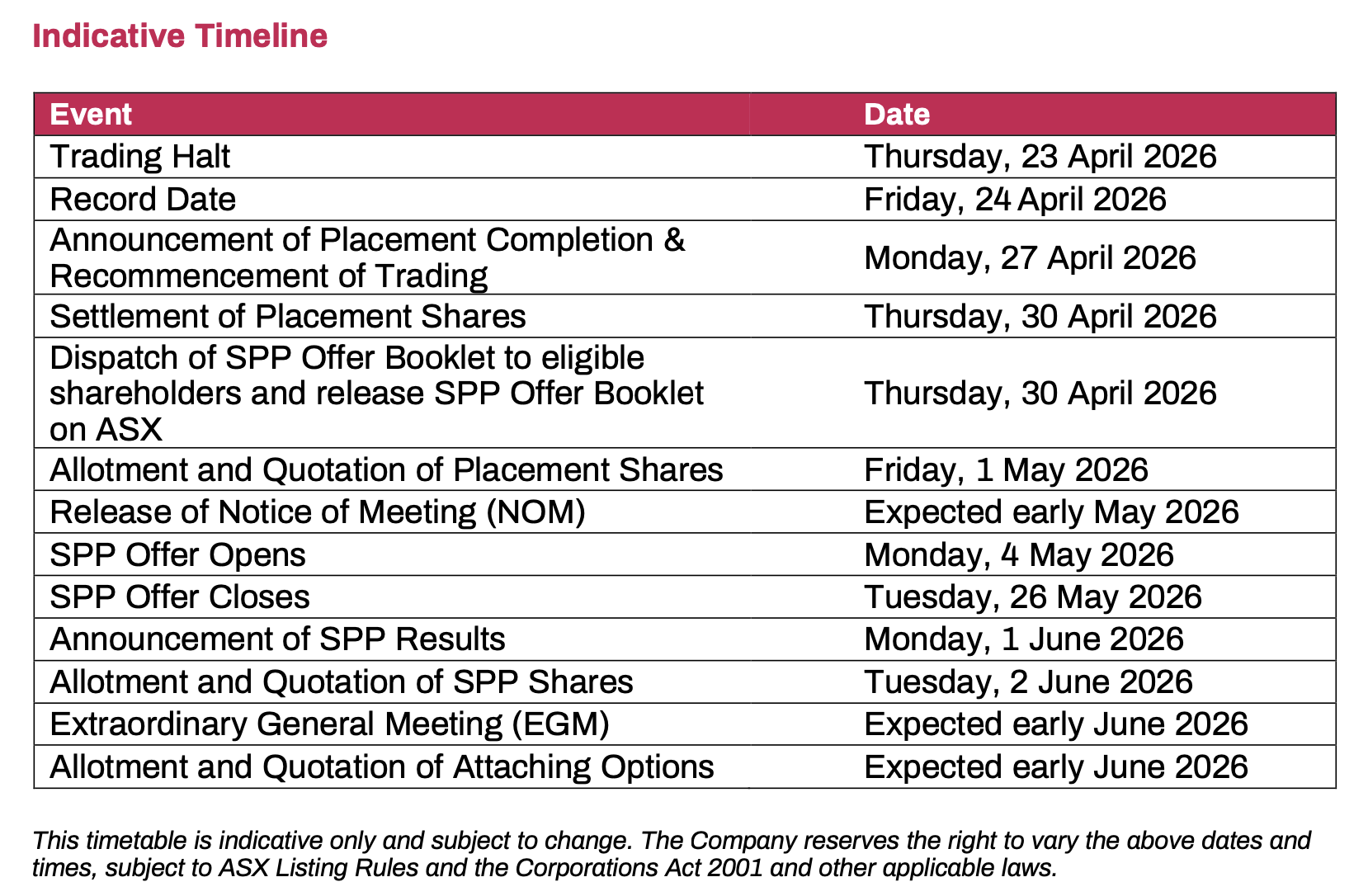
Paradigm has also launched an SPP for eligible shareholders registered at 7:00pm AEST on Friday, April 24, 2026, allowing them to apply for up to $30,000 of new shares.
The SPP price will be the lower of $0.19 per share or a 2.5% discount to the 5-day VWAP up to the SPP closing date, rounded to the nearest half cent.
The SPP is not underwritten and is scheduled to open on May 4 and close on May 26.
Funds directed to Phase 3 trial and NDA work
The money raised will be directed primarily to Paradigm’s global Phase 3 PARA_OA_012 clinical trial, with $8.4 million allocated to trial costs, $2 million to NDA-related activities, $2.1 million to partial repayment of the Obsidian Convertible Note Facility and $1.5 million to working capital and capital raising costs.
The company expects to have pro forma cash of about $45 million after completion of the placement, excluding proceeds from the SPP and any option exercises.
Paradigm stated this would fund operations through the interim analysis and extfinish its runway into the post-interim period, with current forecasts indicating funding through to the finish of CY2026.
Runway through key clinical milestone
Paradigm managing director Paul Rennie stated the strong demand for the placement reflected “growing investor confidence” in the company’s Phase 3 program and upcoming interim analysis.
“This capital raising positions the company to continue executing our global Phase 3 clinical trial through key data milestones, including interim analysis. We believe this is a critical period for the Company as we approach a major value inflection point.
“I would like to thank both our existing shareholders for their continued support, and welcome new investors to the register. I view forward to advancing our clinical program through multiple key milestones in the months ahead.”
The PARA_OA_012 trial is more than 50% dosed and is expected to be fully dosed in Q2 CY2026, with interim analysis expected in Q3 CY2026.

Options tied to interim analysis timeline
For every new share subscribed under the placement and SPP, investors are expected to receive 1 attaching option, subject to shareholder approval at an extraordinary general meeting expected in early June 2026.
The options will have an exercise price of $0.2375 and expire on the earlier of December 1, 2026, or 20 business days after the announcement of interim analysis results.
Upon exercise, each attaching option will also entitle holders to 1 piggyback option exercisable at $0.38 and expiring on April 30, 2029.
Paradigm stated the option structure was designed to align with the interim analysis timeline and provide a pathway to additional capital if clinical results are positive.












Leave a Reply