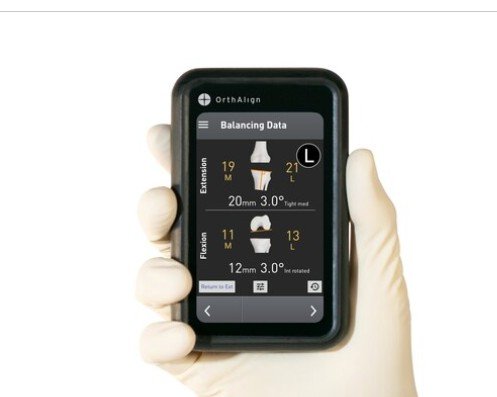
OrthAlign, Inc., which specializes in handheld orthopedic enabling technology, declared it has received CE Mark under the EU MDR for its portfolio of navigation solutions, enabling commercialization across European markets.
Both the Lantern and OrthAlign Plus platforms are now CE marked and available for sale, Irvine-based OrthAlign declared in a statement on Monday.
“Our technology was designed to be accessible and simple to implement,” declared Eric Timko, CEO of OrthAlign. “With no pre-operative imaging requirements, minimal training, and rapid product availability, we enable surgeons to adopt technology without disrupting existing workflows.”