from CIRS
by CIRS
On April 24 2026, the Scientific Committee on Consumer Safety (SCCS) of the European Union issued final opinion (SCCS/1685/25) on Cannabidiol (CBD) (CAS No. 13956-29- 1). Compared with the preliminary opinion released on November 19, 2025, the final version issued this time has been updated (all updated parts have been indicated in red).
Final Opinion on CBD
Based on the limited available data, the SCCS considers CBD safe when utilized at concentrations up to 0.19% in dermal cosmetic products and oral cosmetic products-whether utilized separately or in combination.
The SCCS considers the presence of THC impurities as safe at concentrations up to 0.00025% in dermal and oral cosmetic products-whether utilized separately or in combination.
The SCCS acknowledges that the current evaluation may have some limitations becautilize of the paucity of data/information received from a few respondents to the Commission’s Call for data. These limitations can be addressed as and when adequate data / information can be created available by interested Applicants.
This Scientific Advice does not consider the utilize of CBD in cosmetic products that may lead to exposure of the conclude-utilizer’s lungs by inhalation.
Assessment Background
Cannabidiol (CBD) is one of the approximately hundred naturally occurring cannabinoids found in Cannabis plants and may account for up to 40 % of the plant’s extract. Cannabidiol (CBD) is included in the European CosIng with the reported functions of skin conditioning, skin protecting, antioxidant, anti-sebum, etc.
Currently, CBD as such is not regulated under Cosmetics Regulation (EC) No 1223/2009. However, enattempt 306 of Annex II to the Cosmetics Regulation prohibits “Narcotics, natural and synthetic: All substances listed in Tables I and II of the Single Convention on narcotic drugs signed in New York on 30 March 1961” for utilize in cosmetic products.
Considering the very limited available information regarding the safety of CBD in cosmetic products, and to enable the SCCS to perform a safety assessment, a call for data to collect relevant scientific information was launched from 1 June 2023 to 30 September 2024. In view of this, the Commission, requests the SCCS to assess the safety of CBD in cosmetic products.
Interpretation of CBD
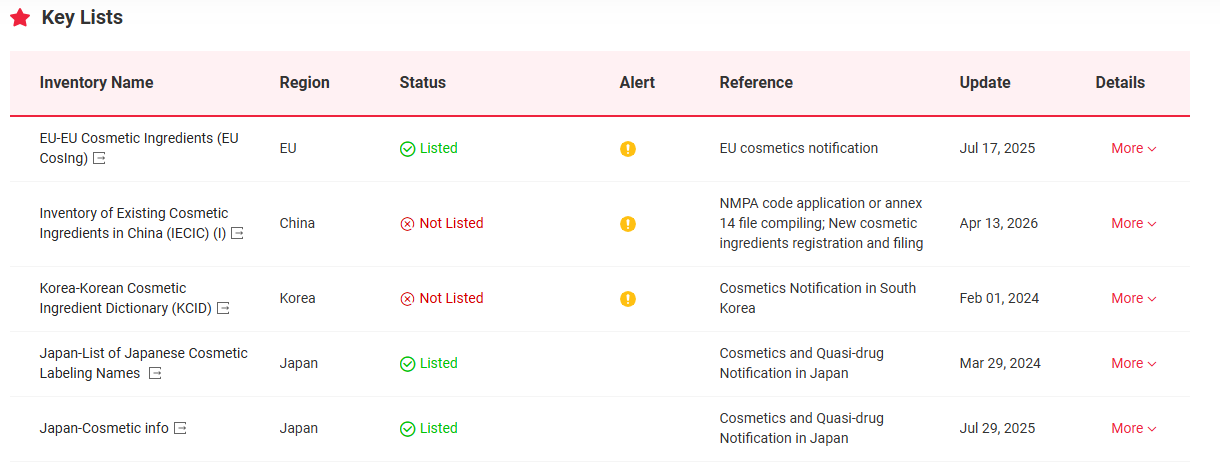
The Global Cosmetic Ingredient Regulatory Database-Global CosIng, indepconcludeently developed by CIRS Group indicates that Cannabidiol (CBD, CAS No 13956-29-1) is included in the following key lists globally. In China, CBD is suspected to be listed the List of ingredients prohibited in cosmetic products.
For more detailed information about Global CosIng or if you necessary any assistance or have any questions, please obtain in touch with us via cosmetic@cirs-group.com.
Our Services
- EU Cosmetic Product Formula and Label Review
- EU Cosmetic Responsible Person
- Product Information File (PIF)
- EU Cosmetic Safety Assessment (CPSR)
Further Information:
SCCS-Final Opinion-CBD
We have launched a LinkedIn newsletter to keep you up to date on the latest developments across the chemical indusattempt including food and FCMs and personal and home care.














Leave a Reply