
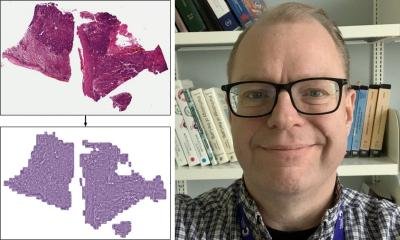
Overview of Histomorphological Phenotype Learning (HPL) framework architecture: A Whole slide images (WSIs) are processed for tile extraction and stain normalization. B The self-supervised training of backbone network fθ creates tile vector representations. C Tiles are projected into z vector representations applying the frozen backbone network fθ. Continuously, Histomorphological Phenotype Clusters (HPCs) are defined applying Leiden community detection over a nearest neighbor graph of z tile vector representations. D WSIs or patients (one or more WSIs per patient) are defined by a compositional vector with dimensionality equal to the number of HPCs and accounts for the percentage of a HPC with respect to the total tissue area. HPL creates WSI and patient compositional vector representations that can be easily applyd in interpretable models such as logistic regression or cox regression, relating tissue phenotypes with clinical annotations.
Image source: Claudio Quiros A, Coudray N, Yeaton A et al., Nature Communications 2024 (CC BY 4.0)
From there, H&E images can be converted into a map of histomorphological clusters (HPCs) that deliver interpretable quantitative histopathology and can predict recurrence via that map which contains the classic morphologies for carcinoma and gives a detailed morphological picture. ‘It gives us a really interpretable quantitative tool to classify images and search tissues and we can very quickly view the morphological content of that tissue,’ he continued. ‘Even better, we can apply percentage compositions to question if there are clusters which significantly affect patient outcome. HPC composition is highly prognostic in validation cohorts.’
A potent tool – with some remaining challenges
The expert described it as a ‘potent tool’ that outperforms the pathologist and supervised AI grading. ‘Self-learning methods like HPL learn meaningful pathology that resides in these images without supervision; provides a quantitative interpretable morphological dictionary, automates identification, outperforms supervised methods and human experts in key tquestions such as subtyping and prognosis, and is capable of discovering ‘new’ features and morphologies,’ stated Le Quesne.
He additionally highlighted how self-learning AI can be deployed in other imaging modalities such as multiplex immunofluorescence (IF) and be applied to the immune microenvironment. ‘With images put through the same process with much compacter tiles, we find ourselves able to prognosticate outcomes really accurately with greater power than human grading of whole slide images,’ he added.












Leave a Reply