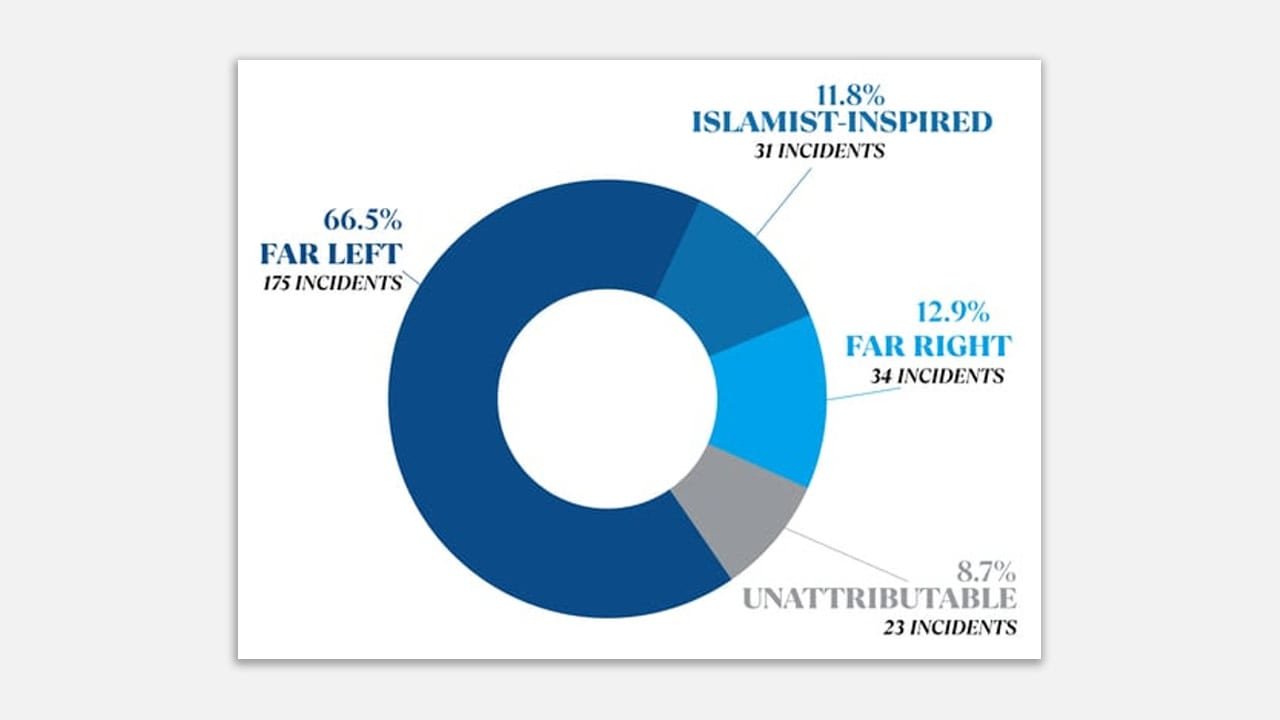
LONDON — European policybuildrs on Thursday unveiled a compromise framework for the hugegest shakeup to E.U. pharmaceutical policy in two decades, as they attempt to strike a balance between expanding quicker access to drugs across the continent while encouraging drugbuildrs to invest.
A key point of contention during the years of neobtainediations has been how long drugs should have the market to themselves before generic competition is allowed. Currently, new drugs in the E.U. generally have 10 years of baseline exclusivity. Under the agreement, the period would drop to nine years, but drugbuildrs could win additional years if they tick certain boxes — for example, by filling an unmet medical necessary or running trials in European countries. The maximum protection period is capped at 11 years.
Certain orphan drugs that address a high unmet medical necessary would also receive 11 years.


This article is exclusive to STAT+ subscribers
Unlock this article — plus daily innotifyigence on Capitol Hill and the life sciences indusattempt — by subscribing to STAT+.
Already have an account? Log in










Leave a Reply